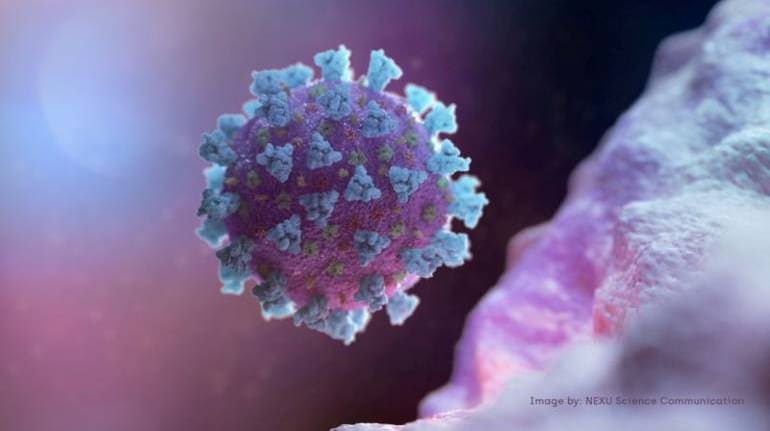
China's only nasal spray vaccine against the coronavirus is expected to start phase I clinical trials in November, and it is recruiting 100 volunteers.
China has approved for trials its first nasal spray vaccine to combat the novel coronavirus that has claimed over 904,000 lives and infected more than 27 million people globally, official media here reported on September 10.
China's only nasal spray vaccine against the coronavirus is expected to start phase I clinical trials in November, and it is recruiting 100 volunteers. It is the only vaccine of its type approved by China's National Medical Products Administration, the state-run Global Times reported.
The vaccine is a collaborative mission between Hong Kong and the Chinese mainland that involves researchers from the University of Hong Kong, Xiamen University, and Beijing Wantai Biological Pharmacy.
Microbiologist from the University of Hong Kong, Yuen Kwok-yung, said the vaccine stimulates the natural infection pathway of respiratory viruses to activate the immune response.
The nasal spray vaccination could generate double protection for vaccine recipients – influenza and the novel coronavirus if it also contains influenza viruses including H1N1, H3N2 and B, Yuen said, adding that it would take at least another year to finish the three clinical trials.
A Beijing-based immunologist told the daily that compared with injections, a nasal spray vaccination is easier to administer and would also be easy to mass-produce and distribute as it adopts the mature influenza vaccine production technology.
The nasal spray vaccine uses live attenuated influenza vaccine; the other four technical routes China is using to develop the coronavirus vaccines are inactivated vaccines, adenoviral vector-based vaccines, and DNA and mRNA vaccines.
The inactivated vaccine is estimated to be the earliest to be in the market, the report said. The immunologist said the new vaccine may not cause systemic side effects, but side effects may occur in the respiratory system such as asthma and shortness of breath.
Ideally, scientists do not expect side effects except for minor nasal obstruction or rhinorrhea, Yuen said. It is not yet clear whether immunity generated from nasal spray vaccinations will last longer than for injected vaccines, the report said.
China has approved three COVID-19 vaccine candidates for clinical trials. It has also authorised emergency usage of COVID-19 vaccines developed by some select domestic companies.
"We've drawn up a series of plan packages, including medical consent forms, side-effects monitoring plans, rescuing plans, compensation plans, to make sure the emergency use is well regulated and monitored," Zheng Zhongwei, head of China's coronavirus vaccine development task force told the official media here last month.
An emergency use authorisation, which is based on Chinese vaccine management law, allows unapproved vaccine candidates to be used among people who are at high risk of getting infected in a limited period.
The Global Times has previously reported that employees of state-owned enterprises (SOEs) preparing to go abroad and the frontline medics have been offered two choices of domestic inactivated vaccine candidates developed by Sinopharm for urgent use.
The novel coronavirus, which originated from China's Wuhan city in December last year, has claimed 904,485 lives and infected 27,902,002 globally. The US is the worst affected country with 190,887 deaths and 6,363,729 infections, according to the Johns Hopkins University data.
Follow our full coverage of the coronavirus pandemic here.

_2020091018165303jzv.jpg)